But otherwise this is how you calculate the average atomic mass of a particular element. So we have to make sure, so pretty much it's pretty much the same thing and that's the reason why it's a slightly different number due to measurements and things like that. Notice that it is slightly different than what we have found in the periodic table, that's okay because our measurements are different and we had to take into account. When you add them all together we get a number of actually I'm going to have to pick up my calculator real quick 39.010 we make sure they're correct number of this one has two so we make sure our decimal place two which is this is and this our atomic mass unit for potassium and so average atomic mass. Now keeping in mind this has four also, so we're going to get rid of one, this guys has three in this case we're going to get rid of this one to make it has three, and this guy has one this guy has one so we're good. 
Now lastly we're going to take the percentage of 0.02 change it to decimal 0.0002 and multiply that by 40 its mass number and we're going to get 0.008. Next we're going to do the same thing for the next guy we're going to change 5.81 to decimal in this case 0.0581 multiplying it by its mass number 41 it's going to give us 2.381. So we're going to say 93.90, we're going to say it's 0.9390 we're then going to multiply it by it's mass number in this case 39 and we're going to get 36.621. Well we have to calculate the average atomic mass and everything, so in this case how we do that is change the percentages to decimal places. These guys are all isotopes of potassium, these are all isotopes of potassium that exist, so still how do we get those numbers to equal 39.098 which is what the periodic table told us. Lastly we have 0.02% of the potassium atom has a mass number of 40 again 19 protons and this case 21 neutrons. 5.81% of potassium atoms that are in analyzed has a mass number of 41, in this case 19 protons which make sense and in this case 22 neutrons different. What that means is, there's 19 protons and 20 neutrons. 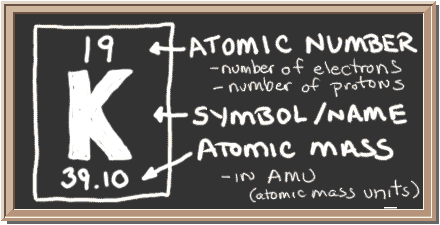
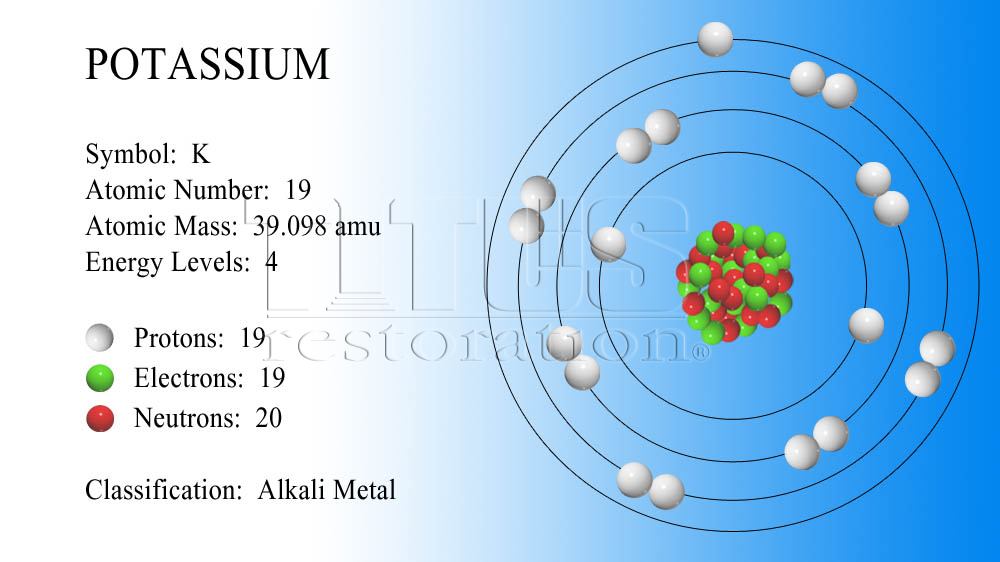
In my banana and in your banana and everybody's banana in the whole world if we pull out all potassium atoms and analyze them we find that 93.90% of potassium atoms in any banana has a mass number of 39. So how do we calculate that? How do we get that answer? Well the bananas are great source of potassium, so we're going to talk about bananas. That is the weighted average of all the isotopes of potassium in the world. So when you average them all together you can get this funny number of 39.098 it's the average atomic mass in atomic mass unit. Well protons and neutrons all have round numbers of 1amu however they have varying numbers of neutrons within every different number of different potassium atoms in the world. So you wonder like why is there a decimal place? We also have a chemical symbol in this case K, K for potassium which doesn't make sense but it's the latin root for potassium, and then we have this funny number at the bottom 39.098 what is that? Well that is the atomic mass of all the potassium atoms that exist. The first thing we have is potassium which is obviously is a chemical name, then we have the number 19 and that atomic mass, sorry our atomic number which means that it has 19 protons for every potassium atom. So when you pull out the periodic table and extension of it we're going to look at potassium for example, we have bunch of information that we have to look through and decipher. So we're going to focus mainly on the protons and neutrons within an element which gives it its mass. So when you think of an atom what gives it the mass, well in the nucleus we have the proton and we also have the neutrons both of 1amu but those have mass surrounding the nucleus we have electrons which is 0amu or essentially weightless comparatively. You will need to refer to a periodic table for proton values.Alright so we're going talk about the atomic mass of an atom and what that actually means. In this notation, the atomic number is not included. It is denoted by ‘mol’ and is also referred to as gram molecular weight. Symbol-mass format for the above atom would be written as Cr-52. 1,90,204 Atoms and Molecules 55,718 Size of an Atom or Ion 81,303 Atoms, Molecules and Ions 31,338 One Mole of a substance is defined as the total number of atoms in 12 grams of Carbon-12 isotope. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
